Management and surveillance
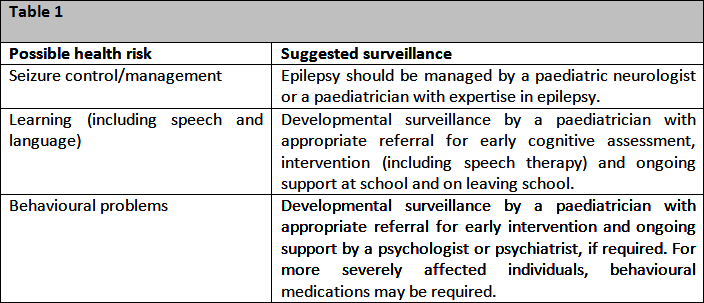
Currently, there is no known cure for CE. Epilepsy should be managed by a paediatric neurologist or a paediatrician with expertise in epilepsy. Early developmental and behavioural diagnosis and intervention with education programs can maximise cognitive and social potential.
Parents should be encouraged to promptly discuss concerns about possible seizures, learning difficulties, or behavioural problems with their child’s paediatrician or paediatric neurologist so that these symptoms can be appropriately diagnosed and managed.
Genetic counselling
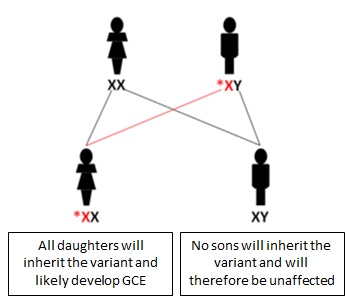
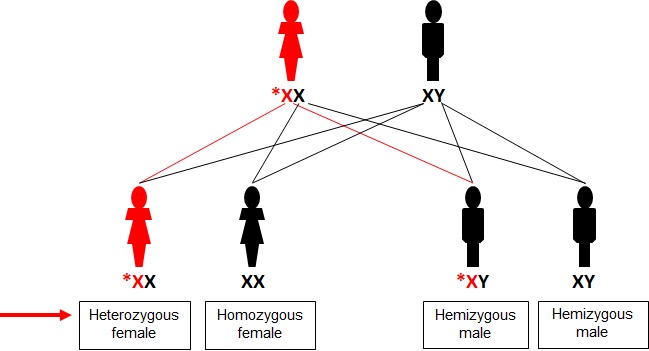
Individuals and their families with a PCDH19 pathogenic variant should be referred to a geneticist and for genetic counselling. This is especially important given the unusual X-linked dominant pattern of inheritance with male sparing. Approximately half of the individuals with CE have a family history of epilepsy. However, the expression pattern is unusual given only females are affected and daughters can inherit the variant from their unaffected fathers or affected or unaffected mothers (Figure 1). The expression pattern in CE is the “inverse” of typical X-linked inheritance, where males are affected and females are unaffected carriers (Figure 2).
Figure 1: X-linked dominant inheritance with male sparing and expression pattern in CE. Note that all daughters of an unaffected (transmitting) male inherit the PCDH19 pathogenic variant and most are affected. A femal with a PCDH19 pathogenic variant will transmit this to 50% of her daughters.
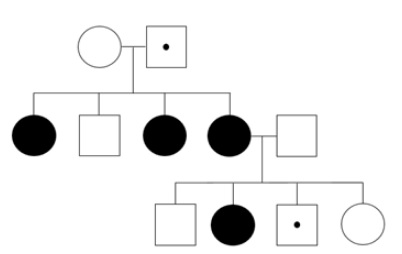
Figure 2: Typical X-linked recessive inheritance
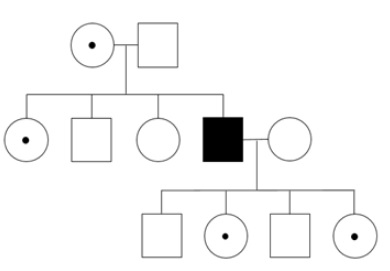
Males inherit an X chromosome from their mother and a Y chromosome from their father. Males who inherit an X chromosome with an abnormal PCDH19 gene are unaffected and are therefore considered transmitting carriers of CE. Females have two X chromosomes; one inherited from their mother and one from their father. Each X chromosome has a PCDH19 gene. However, while females have two X chromosomes in each of their cells, only one X chromosome is generally active. This is due to an evolutionarily old molecular process called X chromosome inactivation (see also below), which prevents the two X chromosomes from being expressed in one cell. Females who have a pathogenic variant in one of their PCDH19 genes are called heterozygotes and are generally affected (90% penetrance).
Around half of the females with CE are sporadic, with no family history of the disorder. The majority of these have a de novo PCDH19 pathogenic variant. However, in a small percentage germline mosaicism can occur where the genetic change occurs later in the parents’ early development. The pathogenic variant is not present in the mother or father’s brain, so they are unaffected. The variant is present in their germline cells. Therefore, the chance that they may have another affected child is lower than for a heterozygous female carrier (50% chance of transmission to daughters) or a transmitting male (100% chance of transmitting to daughters). However, the risk of having another affected child for individuals with gonadal mosaicism is not negligible. Due to the complex nature of CE inheritance, it is important that families are offered comprehensive genetics counselling, especially if they are planning another pregnancy. Their children should also be offered counselling when they grow up. The sons of a mother with a PCDH19 pathogenic variant have a 50% chance of inheriting the pathogenic variant and then a high risk of having affected daughters.
What are the chances of a female with a PCDH19 pathogenic variant having a child with a CE?
Females with a PCDH19 pathogenic variant who are on the mild end of the disease spectrum or asymptomatic carriers may want to have children. Females with a PCDH19 pathogenic variant, regardless of their disease penetrance, have a 50% chance of passing on the X chromosome with the working PCDH19 gene and a 50% chance of passing on the X chromosome with the PCDH19 pathogenic variant (see diagram).
What are the chances of a male (hemizygous or mosaic) with a PCDH19 pathogenic variant having a child with CE?
Males pass on their X chromosome to all their daughters and a Y chromosome to all their sons. A carrier male will pass on the X chromosome with the PCDH19 pathogenic variant to all of his daughters. This means all their daughters will be at high (due to ~90% penetrance) risk of developing CE. As a male with a PCDH19 pathogenic variant passes on his Y chromosome to all his sons, they will not carry their father’s PCDH19 pathogenic variant.